The recalled over-the-counter product contains the regulated substance acetaminophen which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.
About 25,660
Kroger at 800-576-4377 (800-KRO-GERS) from 7 a.m. to 12 a.m. (midnight) ET Monday through Friday, 7 a.m. to 9:30 p.m. ET Saturday and Sunday, online at www.kroger.com/i/recall-alerts, www.kroger.com and click on “Recall Alerts” at the bottom of the page, or www.aurohealth.com and click on “RECALL” at the top of the page for more information.
Recall Details
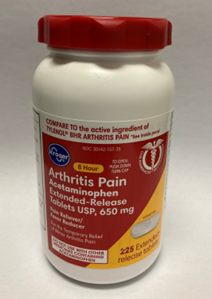
This recall involves the Kroger brand acetaminophen. The red, white, and yellow label states, Kroger, Acetaminophen, Arthritis Pain, Extended-Release, Tablets USP, 650 mg, 225 extended-release tablets. The bottle has a red continuous thread gear closure. UPC number 04126001284 and lot numbers P2100890, P2100891, P2100992 (each with expiration date Aug-2023) and P2101010 (with expiration date Apr-2023) are included in this recall. The UPC number, lot number and expiration date are printed near the drug facts panel on the label on the back of the bottle.
Consumers should immediately store the recalled product in a safe location out of reach and sight of children. Contact Kroger for information on how to properly dispose of the product and receive a full refund.
None reported
Aurohealth LLC, of East Windsor, New Jersey
Note: Individual Commissioners may have statements related to this topic. Please visit www.cpsc.gov/commissioners to search for statements related to this or other topics.
If you are experiencing issues with a recall remedy or believe a company is being non-responsive to your remedy request, please use this form and explain the situation to CPSC.
The U.S. Consumer Product Safety Commission (CPSC) is charged with protecting the public from unreasonable risk of injury associated with the use of thousands of types of consumer products. Deaths, injuries, and property damage from consumer product-related incidents cost the nation more than $1 trillion annually. Since the CPSC was established more than 50 years ago, it has worked to ensure the safety of consumer products, which has contributed to a decline in injuries associated with these products.
Federal law prohibits any person from selling products subject to a Commission ordered recall or a voluntary recall undertaken in consultation with the CPSC.
For lifesaving information:
- Visit CPSC.gov.
- Sign up to receive our email alerts.
- Follow us on Facebook, Instagram, X, BlueSky, Threads, LinkedIn and Truth Social.
- Report a dangerous product or product-related injury on www.SaferProducts.gov.
- Call CPSC’s Hotline at 800-638-2772 (TTY 800-638-8270).
- Contact a media specialist.