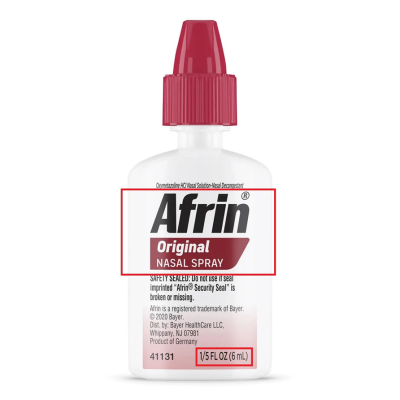
The 6 mL nasal sprays contain an imidazoline, which must be in child-resistant packaging or meet the labeling requirements for non-complying packaging as required by the Poison Prevention Packaging Act. The 6 mL nasal spray’s packaging is not child-resistant nor bears the required labeling statement, posing a risk of serious injury or illness from poisoning if the contents are swallowed by young children.
About 786,100
Bayer toll free at 800 317-2165, Monday through Friday 8 a.m. through 8 p.m. ET, or online through a recall refund webform available at www.livewell.bayer.com/afrin-original-spray-recall. The form can also be accessed by clicking the “Safety Recall Info” button located at the top right of the landing page www.afrin.com, which will take the consumer to the recall information page.
Recall Details
This recall involves unexpired Travel Size Afrin® Original Nasal Spray 6 mL bottles, with Lot numbers 230361, 240822, 241198, 250066, 250152, 250646, and 250831. These travel size bottles have “Afrin® Original Nasal Spray” and “1/5 FL OZ (6 mL)” printed on a label located on the front of the bottle. It is a six-digit number, followed by the expiration date in following format: “YYYYMMM”. No other sizes of Afrin are included in this recall.
Consumers should immediately secure the recalled bottles out of sight and reach of children and go to the webform posted on www.livewell.bayer.com/afrin-original-spray-recall to begin a request for a refund. Consumers will be asked to take and submit a photo of the product before disposing of it in order to receive a refund.
None reported
The U.S. Consumer Product Safety Commission (CPSC) is charged with protecting the public from unreasonable risk of injury associated with the use of thousands of types of consumer products. Deaths, injuries, and property damage from consumer product-related incidents cost the nation more than $1 trillion annually. Since the CPSC was established more than 50 years ago, it has worked to ensure the safety of consumer products, which has contributed to a decline in injuries associated with these products.
Federal law prohibits any person from selling products subject to a Commission ordered recall or a voluntary recall undertaken in consultation with the CPSC.
For lifesaving information:
- Visit CPSC.gov.
- Sign up to receive our email alerts.
- Follow us on Facebook, Instagram, X, BlueSky, Threads, LinkedIn and Truth Social.
- Report a dangerous product or product-related injury on www.SaferProducts.gov.
- Call CPSC’s Hotline at 800-638-2772 (TTY 800-638-8270).
- Contact a media specialist.